Diabetic Monitor Recall Linked to 7 Deaths Sparks Alarm
Abbott has recalled its Freestyle Libre 3 and Libre 3 Plus continuous glucose monitors due to inaccuracies linked to seven deaths and 860 serious injuries. The recall impacts millions who rely on the devices for diabetes management, with experts urging users to have backup monitoring methods.
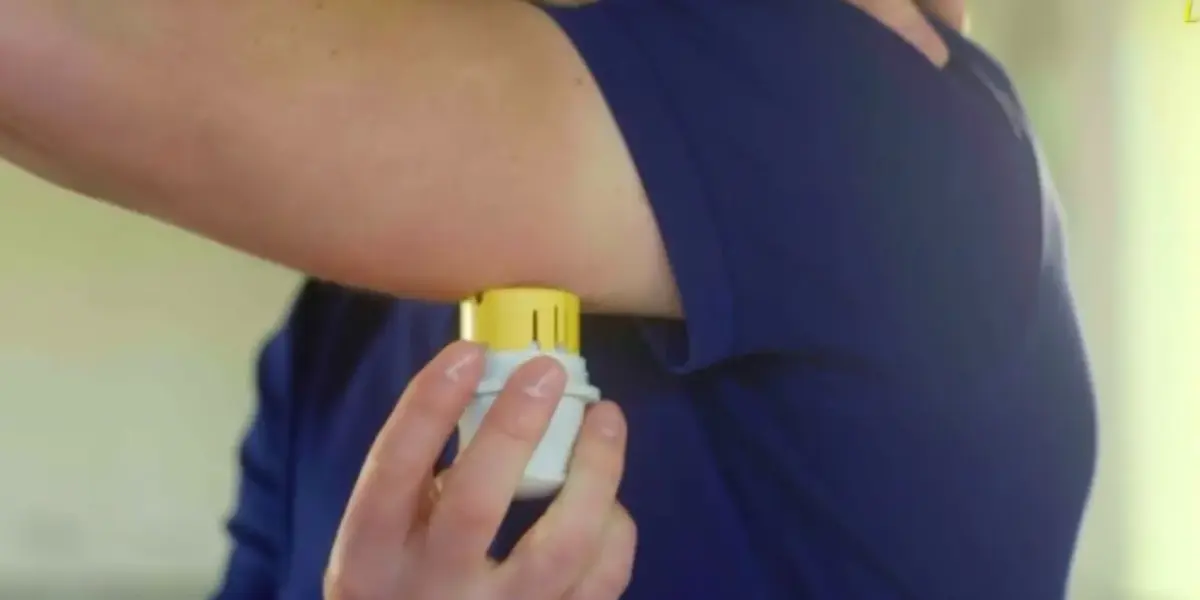
Abbott Recalls Glucose Monitors After Reports of Serious Injury and Death
A significant international recall of Abbott’s Freestyle Libre 3 and Libre 3 Plus continuous glucose monitors (CGMs) has sent shockwaves through the diabetic community. The devices, which provide painless, real-time blood sugar readings via a sensor worn on the arm, are now under scrutiny following reports of inaccuracies that have been linked to at least seven deaths and 860 serious injuries. The recall, announced on November 23rd, 2023, affects millions of users who rely on these devices for daily diabetes management.
A Father’s Devastating Loss
The gravity of the situation is starkly illustrated by the tragic experience of Devonte Ford, a licensed caregiver whose father, Michael, was bedbound due to diabetes. Michael relied heavily on the Freestyle Libre 3 Plus for managing his condition, a stark contrast to the traditional finger-prick methods he used in the past. “What are your memories of him dealing with diabetes?” Devonte was asked. “Always like the finger prick, you know, that was something I always seen him do,” he recalled, highlighting the technological shift in diabetes care.
On the morning of November 16th, Devonte noticed his father’s blood sugar readings on the monitor were unusually low. Despite administering sweetened coffee, a common practice to raise blood sugar, the monitor’s readings remained consistently low, and the readings did not reflect the expected increase. “I thought something was pretty strange with that,” Devonte stated, noting that no amount of carbohydrates seemed to elevate his father’s blood sugar levels according to the device.
As his father’s condition worsened, Devonte observed him speaking softly and made the difficult decision to call 9-1-1. By the time paramedics arrived, Michael was unresponsive. “He had ended up coding, so he had a cardiac arrest and he couldn’t breathe on his own at that point,” Devonte recounted. “So they had to do CPR to him.” Tragically, Michael’s blood sugar level was recorded at 551 – more than eight times higher than what the Freestyle Libre 3 Plus had indicated. Michael never regained consciousness and passed away the following day, November 17th.
Recall and Company Response
Seven days after Michael’s death, Abbott announced the international recall for the Freestyle Libre 3 and Libre 3 Plus sensors, citing incorrect low readings. As of January 7th, the company had received reports of 860 serious injuries and seven deaths associated with the faulty devices. Devonte’s mother discovered the recall notification online, a moment Devonte described as feeling like “divine intervention.” He has since filed a lawsuit against Abbott.
Abbott has not yet responded to the lawsuit but issued a statement to NBC News: “We are deeply saddened by the situation. We are committed to the safety and quality of our products. We take all adverse event reports seriously. Each situation is thoroughly documented, investigated and shared with regulatory authorities as required.”
Expert Advice and Precautions
Dr. Melanie J, Director of NYU Langone’s Obesity Research Program, emphasizes the importance of these devices while also cautioning against blind reliance. “I think these monitors are a great tool, but as with any medical device or any tool, there can be malfunctions and there can be ways that they’re not used properly,” she explained. “And so it’s just not to blindly rely on them.”
Devonte Ford shared that he had never experienced any issues with the monitor prior to the incident. “We just never had a scare ever. And this was the first one and the last one,” he said, his voice filled with the lingering trauma of the event.
FDA Guidance and User Actions
The Food and Drug Administration (FDA) has advised individuals possessing these specific Abbott sensors to cease using them and discard them immediately. Approximately 3 million of these devices were manufactured. For individuals who suspect their glucose readings may be inaccurate or experiencing unusual symptoms, healthcare professionals recommend always having a backup plan.
“Yeah, you always want to have a backup plan. And that backup plan is that fingerprick glucose monitor that you want to have,” reporter Anne Thompson advised. “And you should turn to that if something seems off.” This advice underscores the critical need for vigilance and secondary verification when managing a condition as sensitive as diabetes, especially when relying on technology that has shown potential for critical failure.
Looking Ahead
The recall of Abbott’s Freestyle Libre 3 and Libre 3 Plus sensors raises critical questions about the reliability of medical devices and the protocols in place to ensure patient safety. As investigations continue and legal proceedings unfold, the diabetic community will be closely watching for further developments, demanding accountability and reassurance that such devastating failures will not be repeated. The focus remains on safeguarding the millions who depend on these technologies for their health and well-being.
Source: Diabetics who rely on glucose monitors alarmed by recall linked to 7 deaths (YouTube)





